
We don’t need to rent a convention center when we have an all-team meeting. We don’t have a campus, a cafeteria, or a corporate jet. Instead, we have a connection with each and every one of our employees. Nobody here is a number. Everybody has a name.
Being a smaller organization allows us to move quickly, communicate effectively, and collaborate consistently. It gives us the ability to really dig into the value each team member contributes as a unique individual in respect to their work, and to embrace diversity, equity, and inclusion on the most human level possible. These perspectives allow us to generate meaningful solutions that create better patient outcomes. We value what everybody has to say.
At Viscus Biologics we recognize that sometimes a full-time job doesn’t fit your life, but you fit into our team. Because of that, we’re happy to offer full-time and part-time roles so you can enjoy your own quality of life while helping create it for others.
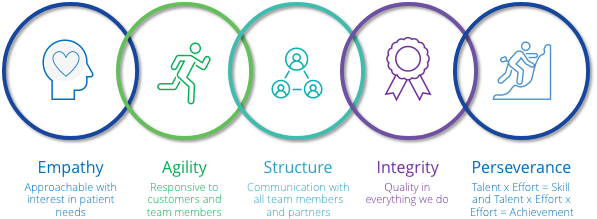
The culture at Viscus Biologics emanates from core values shared by team members throughout the organization.
Viscus Biologics is committed to attracting and retaining the best talent available. Employees receive the following benefits:


Internship and Co-Op Disciplines:
Requirements for a Co-Op:
Requirements for an internship:
Viscus Biologics believes that a drug-free workplace is essential for the productivity and safety of our employees and the protection of our business. Therefore, we are committed to providing a drug-free working environment for all employees. All applicants who accept an offer of employment will be required to submit to drug testing as a condition of employment.
The best way to apply is to view all open positions here. Select the role (or roles) you're interested in, click the “Next Page” button, and follow the instructions to complete an online application. Please upload your resume when asked.
Viscus Biologics use a tool called The Culture IndexTM for our application process. This allows us to match your traits and strengths to the requirements of the position. It helps us to ensure the right people are in the right positions and are a fit for our company culture. The baseline of a great culture is that people get to come to work at be themselves!.
We’re looking for ambitious individuals with an eye for quality who are keen to develop a career at Viscus Biologics. You should demonstrate a genuine interest in the role you’re applying to and be able to explain why you’re applying for this opportunity with us specifically.
Once you have applied and your application has been reviewed, you’ll be invited to a call with a member of our Human Resources team. After that, there will be one or more interviews (phone, virtual, or in-person). All these interviews act as opportunities for us to get to know you and vice versa. The number of interviews you go through and the number of stakeholders you talk to may vary by position. Technical skills assessments may also be used as part of the process.
Viscus Biologics is committed to working with and providing reasonable accommodations to individuals with disabilities. If you require assistance or an accommodation because of a disability to participate in the application process, please fill out the contact form below.
You may receive help with relocation if eligible. We understand that it’s a big challenge whether you’re moving alone or with a family, so if relocation is required, our goal is to make it as stress-free as possible. Relocation benefits vary by role, so ask your recruiter for details.
Viscus Biologics is an equal opportunity employer and all qualified applicants will receive consideration for employment without regard to actual or perceived race, color, creed, religion, national origin, ancestry, citizenship status, age, sex, or gender (including pregnancy, childbirth, lactation, and related medical conditions), gender identity or expression (including transgender status), sexual orientation, marital status, military service and veteran status, disability, protected medical condition as defined by applicable state or local law, genetic information or any other characteristic protected by applicable federal, state or local laws and ordinances.



